Understanding the Significance of FDA 21 CFR Part 11 in the Pharma Industry

Overview of FDA 21 CFR Part 11
FDA 21 CFR Part 11, titled “Electronic Records; Electronic Signatures,” establishes guidelines for the use of electronic records and electronic signatures in the pharmaceutical industry. It aims to ensure the authenticity, integrity, and confidentiality of electronic data generated in drug manufacturing, clinical trials, and other regulated activities. Compliance with Part 11 is vital for maintaining data accuracy, preventing fraud, and meeting the regulatory expectations of the Food and Drug Administration (FDA).
Key Requirements of FDA 21 CFR Part 11
- Electronic Records: Part 11 mandates that electronic records be secure, reliable, and accurate throughout their lifecycle. This includes requirements for electronic documentation of manufacturing processes, batch records, laboratory results, and other critical data.
- Electronic Signatures: Part 11 permits the use of electronic signatures as legally binding alternatives to traditional handwritten signatures. It sets guidelines for the use of cryptographic controls to ensure the authenticity and non-repudiation of electronic signatures.
- Audit Trails: Part 11 requires systems to have audit trails that capture a chronological record of all system activities, including user actions, data modifications, and system events. These audit trails aid in traceability, accountability, and data integrity verification. Docuply as a document management system for the pharmaceutical industry offers an audit trail by default.
Benefits of Compliance
Compliance with FDA 21 CFR Part 11 offers several benefits to the pharmaceutical industry. It ensures data integrity, reducing the risk of errors, tampering, and fraudulent activities. Additionally, it enhances patient safety by maintaining accurate and reliable records throughout the drug development and manufacturing process. Compliance also streamlines regulatory inspections and audits, as organizations adhering to Part 11 are better prepared to demonstrate data accuracy and compliance with regulatory requirements.
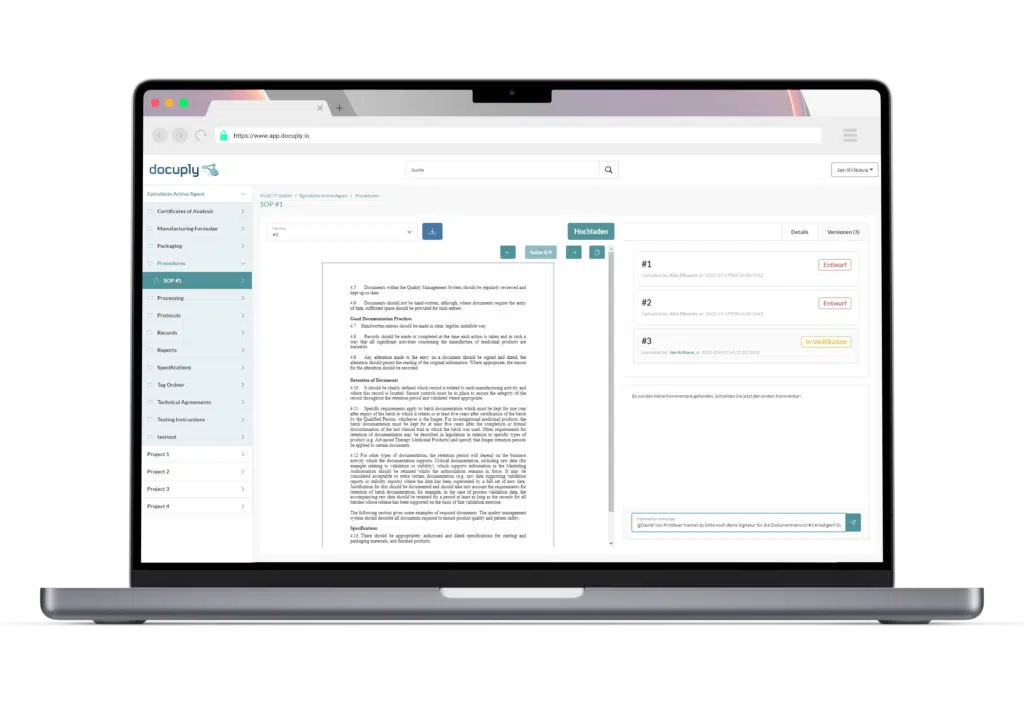
With Docuply, you and your third parties can move smoothly and GLP-compliant from preclinical studies to clinical trials.
Challenges in Implementation
Implementing FDA 21 CFR Part 11 can pose challenges for pharmaceutical companies. Organizations must invest in robust electronic systems and software solutions that meet the stringent requirements outlined by the regulation. Additionally, training employees on proper system usage, data security, and electronic signature protocols is essential. Maintaining compliance in a rapidly advancing technological landscape also necessitates regular updates, system validations, and ongoing monitoring.
Conclusion
FDA 21 CFR Part 11 plays a vital role in ensuring data integrity, security, and compliance in the pharmaceutical industry. By adhering to the regulation’s requirements for electronic records, signatures, and audit trails, pharmaceutical companies can protect patient safety, enhance operational efficiency, and maintain regulatory compliance in an increasingly digital era.
Test all the advantages of a digital document management system specially developed for the pharmaceutical and biotech industry now without obligation. You can get started in just five minutes, see for yourself.
Quality management in the pharmaceutical and biotech industry: Current challenges in the cooperation between pharmaceutical companies and CROs, CMOs and CDMOs